It may sound cliched, but it’s true: Every cloud does have a silver lining.
While the period of Home-Based Learning (HBL) was a challenging time for teachers, students and their parents as we had to quickly transition to taking classes online, it also provided us with a good opportunity to rethink our approach to our lessons to make them more engaging.
As a Chemistry teacher, what worried me was the lack of access to physical science laboratories during HBL. Did that mean that my students could no longer carry out science experiments?
Taking experiments online
The week before HBL kicked in, I had just started teaching the topic of electrolysis – generating a chemical reaction by passing electric current through a substance – to my Secondary 4 students.
While students are not tested on electrolysis in their practical exam, they are expected to understand and apply the theory in their written exam. So, they would conduct mini-electrolysis experiments in the lab to reinforce their understanding of the concept. With HBL, this would not be possible.
So, I started to ponder:
“How do I show students how electrolysis works when they are learning from home?
“How do I let students discover what happens through the electrolysis of different substances?”
“Will my students lose out in terms of learning if they do not have the chance to conduct a practical lesson in electrolysis?”
The answer lay in virtual experiments that were available on the Student Learning Space (SLS). When the students hopped on to the electrolysis lesson on the SLS, they could access a simulated experiment. There is a 3-D model of an electrolysis set-up, where students could alter various aspects of an electrolytic cell.
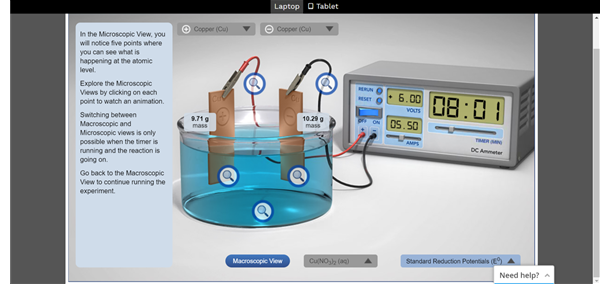 To start off, they could select the type of electrode metals for the experiment, for example, copper or zinc. They could also tweak the settings of the battery and with a virtual ammeter – a device to measure electric currents – measure the different results.
To start off, they could select the type of electrode metals for the experiment, for example, copper or zinc. They could also tweak the settings of the battery and with a virtual ammeter – a device to measure electric currents – measure the different results.
The virtual experiments enabled students to view what happens to each electrode at the atomic level. This is something that is not possible in a physical lab.
At the end of the virtual experiments, students would compare their observations with their classmates and have a mini discussion on SLS.
I would also follow up by relating these experiments to real-world examples. I explained to them how electrolysis was crucial to the development of lithium ion batteries in their smartphones.
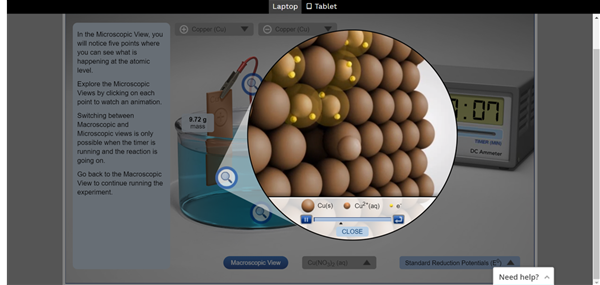
While it would have been nice to see the actual chemical reaction in the real lab setting, these virtual experiments provided an accurate enough simulation to help students appreciate and gain a deeper understanding of electrolysis.
I am glad that I managed to teach my students the fundamentals of the topic in class right before HBL kicked in! This way, they were familiar with the terminologies and knew what variables to take note of when they were conducting the online experiments in their own homes.
Sparking curiosity
I believe these virtual experiments helped my students become more self-directed in their learning. They tried the experiments on their own time and given this space to explore, they became more curious about how each variable affected the outcome of the experiment.
Perhaps it was because it was easier to tweak the variables and observe the outcomes in these virtual experiments, and therefore it was more exciting to them. It was good to see my students enthused about this, and I hope that this interest helps them retain the information better and apply it in their written exam.
Back to school
Having said that, there were challenges to virtual classes as well.
For one, not every student had been diligently following the online classes – so there was inconsistent mastery of the topic among them. Keeping track of their work and giving each of them feedback also took more time.
 Once back in school, I had to remind students about what they had missed out and why it was important for them to go back and revisit the lessons. This was especially crucial as they were taking their ‘O’ levels this year.
Once back in school, I had to remind students about what they had missed out and why it was important for them to go back and revisit the lessons. This was especially crucial as they were taking their ‘O’ levels this year.
So, while I am thankful that tech tools made science lessons more interactive and interesting, there’s nothing like talking to my students in person.
You form closer bonds with them and it makes class time a lot livelier.








